Freshwater mussels found in Bear and Cottage Lake Creeks during habitat assessments in 2001
In conjunction with the Sammamish-Washington Assessment and Modeling Program
April 15, 2002
Karen Fevold, King County Water and Land Resources Division
Jennifer Vanderhoof, King County Water and Land Resources Division
Download the full report here.
Summary and Introduction
King County conducted stream habitat assessments in 2001 in Bear Creek and Cottage Lake Creek to characterize the habitat quality, primarily for salmonids. During this habitat assessment, notations were made of freshwater mussel locations, numbers, size, and mussel-bed substrate. This report is intended as a preliminary reconnaissance of the mussel presence in the Bear Creek Basin.
Introduction
King County conducted habitat assessments in 2001 in Bear Creek and Cottage Lake Creek and two of their tributaries to characterize the habitat quality, primarily for salmonids. During this habitat assessment, notations were made of freshwater mussel locations, numbers, size, and substrate. These evaluations of freshwater mussels were intended to be a preliminary reconnaissance only; mussel counts are sometimes estimates, and lengths are typically estimates.
The Bear Creek Basin is located immediately to the east of Little Bear Creek and drains to the Sammamish River, which ultimately drains to Lake Washington. The upper reaches of the Bear Creek Basin still have some large good-quality wetlands and high-quality salmonid spawning and rearing habitat, as well as one of the largest populations of freshwater mussels found in the Puget Sound Lowlands (Toy 1998).
Mussel Diversity
The United States has the richest diversity of freshwater mussels in the world, with 297 different species (Turgeon et al. 1988). Of these 297 species, approximately 72 percent are considered endangered, threatened, or of special concern (Williams and Neves 1995). Sixty-nine species are listed by the U.S. Fish and Wildlife as endangered or threatened, and 21 are endangered and presumed extinct.
Most of the species in the United States live in the southeast, which is home to the richest collection of freshwater mussels in the world. In western Washington, there are three extant species of freshwater mussel: western pearlshell (Margaritifera falcata), Oregon floater (Anodonta oregonensis), and western ridgemussel (Gonidea angulata). Relatively little is known about these species compared to mussels east of the Mississippi River (Toy 1998).
The most common mussel in the Lake Washington watershed is Margaritifera falcata. Observations of Margaritifera falcata in an area of northern Big Bear Creek have been reported to have up to 164 mussels per 0.5 m2 (Rensel 1992). Western pearlshell (Margaritifera falcata) live in rivers and streams with cool, flowing water and sand/gravel substrates.
Mussel Life History and Habitat
Freshwater mussels have two life-history stages, parasitic and stationary. Adult mussels are stationary. They stay in one spot, bury their anterior end in the river bottom, and leave the posterior end and two siphons exposed. They continuously pump water through their bodies. The water enters through the incurrent siphon and exits via the anal siphon. The mussel uses its gills to filter oxygen and food from the water. Their food consists of plankton and other organic matter suspended in the water.
In the spring when mussels are ready to reproduce, the males release sperm into the water, and the females draw the sperm in through the incurrent siphon. The ova are fertilized internally. If mussels aren’t grouped fairly closely to one another, reproduction is hard to achieve. After fertilization, the female holds up to several thousand eggs at a time in her gills, where they can obtain oxygen and have a place to brood until they develop into glochidia, the larval stage of mussels.
In the late spring or early summer, the glochidia are expelled into the water. To survive they must attach themselves to the gills of a host fish, usually a salmonid, within a couple days. Most freshwater mussels parasitize only one type of fish: mussel species in the Pacific Northwest favor salmonids. Without salmon as hosts, mussels cannot successfully reproduce.
Once the larval mussels attach to the fish, the fish’s body reacts to cover them with cells that forms a cyst. The glochidia remain in the cyst for 2 to 5 weeks (depending on the water temperature). After the mussels begin to resemble adults, they break out of the cyst and fall to the bottom of the stream. They fall wherever the fish happens to be and bury themselves in the stream bottom. Only about 10 glochidia out of every million produced actually succeed in infecting a suitable hose (Bauer 1988). To offset this high mortality rate, mussels live long reproductive lives and produce vast numbers of glochidia each year (Bauer 1987). Some species of mussel can live over 100 years. There have been individuals of Margaritifera falcata found to exceed ages of 90 years in Bear Creek (Toy 1998).
Mussels are very sensitive to the quality of their instream habitat and are often thought of as good indicators of the health of a stream, because healthy mussel populations require clean gravel and sand (Moorkens 1999 in Arujo and Ramos 2000). Clean substrate is very important for juveniles because if the substrate becomes clogged with silt, the mussels can no longer obtain oxygen. The mussels have to filter the water where they land as larvae, and so they ingest whatever is around them. They are therefore sensitive to toxins and pollutants in the stream. Mussels require the same clean, cool, oxygenated water that salmon need (in addition to needing the salmon themselves).
Methods
The mainstem of Cottage Lake Creek and the mainstem of Bear Creek upstream from the confluence with Cottage Lake Creek were assessed for instream habitat quality. Parts of Cold Creek and Struve Creek were also assessed. As assessment staff walked in an upstream direction to evaluate salmonid habitat, data on freshwater mussels were also recorded. Data collected include mussel bed location, mussel count, mussel lengths, and the substrate the mussels were found in.
Access to property was obtained before stream reaches were entered. Approved property access often determined which areas were surveyed. The streams were divided into assessment segments (Map 1) based on the Salmon and Steelhead Habitat Inventory and Assessment Project (SSHIAP) stream segment definitions. SSHIAP segments are based on gradient and confinement. All of the mussel data are summarized on a segment basis.
Location
Anywhere one or more mussels were found was considered a mussel bed. The location of the mussel beds was noted either by the hipchain distance from the start of the assessment reach, or using a Trimble Pro-XR or Geo-Explorer 3 GPS unit.
Count and Size
The number of mussels in a mussel bed were either counted or estimated. When the number of mussels was approximately fewer than 20, the mussels were counted; when the number of mussels present appeared to be greater than 20, counts were estimated.
Average size of mussels in a bed was also estimated (large, medium, small). Mussels were occasionally measured (1) if the size category was in question and (2) to calibrate ocular estimations. Large mussels were generally 80-120mm long, medium mussels were 40-80mm, and small mussels were under 40mm.
Substrate
Dominant substrate of the mussel beds was recorded. Substrate type was broken into four categories: medium to coarse gravel (8-64mm), fine gravel (2-8mm), sand (0.5-2mm), and silt (less than 0.5mm).
Results and Discussion
Map 1. Mussel Locations in the Bear Creek Basin All mussels observed are presumed to be Margaritifera falcata. Stream segment data and mussel frequency data are summarized in Table 1. Segment lengths ranged from 363 meters to 1,796 meters. Segment BC2 was the shortest segment at 303 meters, but it was not assessed because property access was not granted for enough of the stream to make a valid habitat assessment. A total of 17 stream segments were surveyed, and M. falcata were found in 10 segments. No mussels were found in Cold Creek or Struve Creek. In Bear Creek and Cottage Lake Creek, 4 segments out of 14 surveyed did not have mussels present. Each of the 4 segments in which no mussels were found had less than 40 percent of the segment length surveyed.
Of the 10 segments in which M. falcata were found, 6 SSHIAP segments ranked 0-1%U (less than 1 percent gradient, unconfined channel). The remaining 7 segments with mussels observed was in the 1-2%U category (1-2 percent gradient, unconfined channel).
Table 1. The percentage of each stream segment surveyed, the SSHIAP ranking of each segment, and mussel bed and mussel frequency (Bear Creek=BC; Cottage Lake Creek=CLC; Cold Creek=CC; Struve Creek=SC).| Segment | Segment length (m) | % Segment Surveyed | SSHIAP ranking | Meters assessed | Mussel beds/km | Mussels/km |
|---|---|---|---|---|---|---|
| BC1 | 894.9 | 94.3 | 1-2% U | 843.9 | 22.9 | 1102.0 |
| BC2 | 303.3 | 0.0 | 0-1% U | 0 | NA | NA |
| BC3 | 1590.1 | 84.2 | 1-2% U | 1339.3 | 1.5 | 6.0 |
| BC4 | 1323.8 | 30.4 | 0-1% U | 525.7 | 0 | 0 |
| BC5 | 692.5 | 47.4 | 1-2% U | 328.2 | 3.1 | 3.1 |
| BC6 | 1678.5 | 93.6 | 0-1% U | 1571.8 | 0.6 | 1.3 |
| BC71 | 1796.2 | 36.6 | 0-1% U | 657.0 | 0 | 0 |
| CLC1 | 669.0 | 15.0 | 0-1% U | 100.0 | 0 | 0 |
| CLC2 | 1161.3 | 54.6 | 0-1% U | 633.6 | 1.6 | 3.2 |
| CLC3 | 363.6 | 39.2 | 1-2% U | 142.5 | 21.1 | 119.3 |
| CLC4 | 612.3 | 58.6 | 1-2% U | 358.7 | 8.4 | 58.5 |
| CLC5 | 953.7 | 39.0 | 1-2% U | 371.9 | 26.9 | 519.0 |
| CLC62 | 421.2 | 117.6 | 1-2% U | 495.3 | 36.3 | 565.3 |
| CLC7 | 928.7 | 72.0 | 0-1% U | 669.0 | 74.7 | 872.9 |
| CLC8 | 768.1 | 39.5 | 0-1% U | 303.2 | 0 | 0 |
| CC 1 | 405.7 | 51.6 | 0-1% U | 209.5 | 0 | 0 |
| CC 2 | unknown | unknown | (not ranked) | 492.0 | 0 | 0 |
| SC 2 | 322.2 | 70.4 | 2-4% C | 226.8 | 0 | 0 |
1 One live mussel was observed while walking downstream to the survey start point, but this mussel was not observed during the actual habitat survey.
2 Over 100 percent of stream segment CLC6 is recorded as having been surveyed. This seeming anomaly results from the inaccuracies of the geographic information system data layer used to map the SHHIAP point versus hipchain length in real time. It is possible that the surveyors continued surveying into the next segment inadvertently because in the field it is not possible to know exactly when one segment ends and the next begins. Mussels per kilometer and mussel beds per kilometer are accurate as reported.
Location
Mussel locations found in the assessed segments in Bear Creek and Cottage Lake Creek are documented on Map 1. Mussel beds in segments BC1, CLC5, CLC6, and CLC7 were generally found throughout the entire assessed area. This corresponds with the higher number of mussels observed in these segments. Mussel beds in segments CLC2, CLC3, CLC4, and BC5 were found sporadically or only in one location in the entire segment.
Two active mussel beds were found in segment BC3 and were located within 50 meters of one another. In BC3, 11 relict mussel beds were found and appeared identical to live beds except that only the shells were remaining. The shells were intact and in the typical upright position in the substrate. Mussel shells were occasionally found throughout Bear Creek and Cottage Lake Creek; however, BC3 was the only segment where the shells in the beds were undisturbed. These findings indicate that the mussels died in situ and not from predation, for example. Only data from live mussels/mussel beds are reported here; however, Map 1 illustrates locations of both live and dead mussel beds.
Count and Size
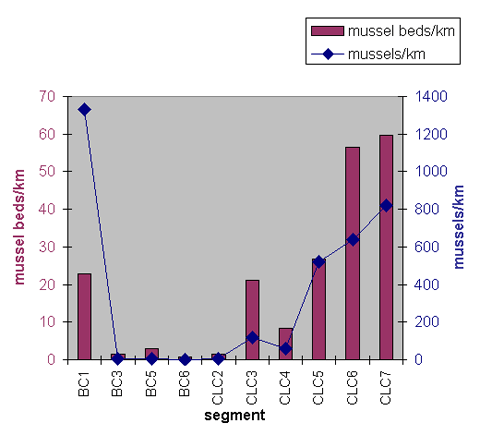
The number of mussels per bed ranged between 1 and 58.1 (Figure 1). Segment BC1 had an average of 58.1 mussels per bed. This number is an outlier; no other segment averaged more than 19.3 mussels per bed. Segment BC1 contains the same reach Toy studied in 1995 and found a mussel density of over 55 mussels per m2. Segment BC1 had by far the greatest frequency of mussels of all the segments in the King County study. In addition, the numbers for segment BC1 reported in this study are probably an underestimate, because on numerous occasions staff exited the stream to avoid stepping on mussels in dense mussel beds.
A study in Ireland determined the minimum population sub-unit of Margaritifera falcata to be 500 reproductive individuals per 0.5 km of river (Moorkens 1999 in Arujo and Ramos 2000). In Bear Creek, Toy (1998) determined sexual maturity was achieved between 9 and 12 years and is dependent on size of the individual mussel. If the minimum population sub-unit in the Pacific Northwest is assumed to be similar to that of Margaritifera falcata in Ireland, only one area in this study currently has a self-sustaining population of mussels—segment BC1 with approximately 1,330 mussels/km. The other three segments with a relatively large number of mussels had frequencies ranging from 519 to 819 mussels/km.
All other stream segments surveyed had mussel frequencies ranging from 6 to 119 mussels/km. If the study in Ireland (Moorkens 1999 in Arujo and Ramos 2000) is indicative of population requirements in this Bear Creek system, the low numbers in the Bear Creek system are probably insufficient to maintain mussel populations over time. It is possible that stream conditions and population ecology in the Ireland study were vastly different from those found in Bear and Cottage Lake creeks.
Mussel sizes were almost always large, although instances of small and medium mussels were recorded. The overall consistency of large mussel size found during the King County habitat study corroborates Toy’s (1998) finding that age distribution of M. falcata in this section of Bear Creek is skewed towards older individuals, with maximum ages exceeding 90 years. The numbers of mussels represent total count, not just reproductive individuals. Because the age structure in Bear Creek is skewed towards older adults, it may be assumed that most or all of the counted mussels are of reproductive age. If BC1 is the only population and source of recruitment stock for the rest of the system and it is found in subsequent studies to have a low recruitment and high mortality rate (Toy 1998), the age structure would indicate a trend toward extinction (Bauer 1987)
 |
|
Figure 1. Frequency of mussel beds/km and mussels/km in each assessment segment in Bear Creek and Cottage Lake Creek. Average number of mussels per bed is reported in the inset table.
Substrate
The composition of mussel bed substrate varied widely in Bear Creek and in Cottage Lake Creek (Figure 2). Mussel beds in segment BC1 showed the most variety; they had all possible substrate types present for at least 10 percent of the area. Over 30 percent of the mussel beds in segment BC1 was silt, and over 40 percent was medium to coarse gravel. Mussels in segment BC3 were predominantly located in fine gravel, and in segment BC5 all the mussel beds were in sand. Segment BC1 had far more mussels and mussel beds present than BC3 or BC5.
In Cottage Lake Creek, segments CLC 5, CLC6, and CLC7 were the only three segments with mussels that had mussel beds located in at least three substrate types. The greatest numbers of mussels found in Cottage Lake Creek were in segments CLC5, CLC6, and CLC7, and the greatest number of mussel beds were found in CLC6 and CLC7.
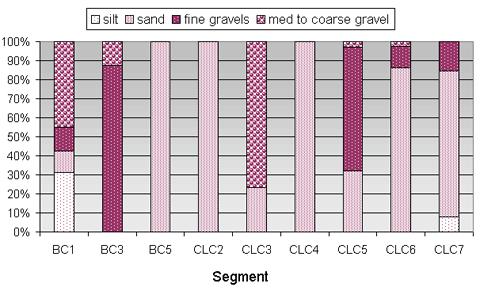
Figure 2. Substrate types found in mussel beds in Bear Creek and Cottage Lake Creek surveyed segments. (No substrate data were collected in segment BC6.)
At least some of the mussel beds were located in sand in each Cottage Lake Creek segment. All mussel beds in two segments (CLC2 and CLC4) were located in sand, and at least 77 percent of the mussel beds in two other segments were located in sand (CLC6 and CLC7). Mussel beds in segment CLC3 were predominantly located in medium to coarse gravel (76 percent) and in CLC5 were predominantly located in fine gravels (65 percent). Most or all of the remaining mussel beds in each of these segments were located in sand. Of the four segments that had the lowest numbers of mussels and mussel beds present, the substrate in mussel beds in three of those segments was 100 percent sand.
Freshwater pearl mussels are at their most vulnerable during the juvenile stage in their life cycle (Bauer 1983). Juvenile M. margaritifera, a species closely related to M. falcata, depend on well aerated coarse sediment. Buddensiek (1991 in Buddensiek et al. 1995) found juvenile mortality of M. margaritifera to be 100 percent within 2 weeks in sand. Toy (1998) found dead M. falcata juveniles in "sand substrate consisting of silt or mud." Toy found a relatively small amount of juvenile habitat in her study area, but almost no juveniles were found. It is possible, then, that the variability in where mussel beds were located may not solely be a result of the type of substrate, but it may also be a result of stream sedimentation. Sedimentation may also be a cause of widespread mussel death (for example, the dead mussel beds in segment BC3).
Acknowledgements
Many thanks to Jessica Kuchan, the tireless field assistant, who worked on all aspects of this data collection effort.
References
Arujo, R., and A. Ramos. 2000. Action plan for Margaritifera margaritifera in Europe. Convention on the conservation of European wildlife and natural habitats.
Bauer, G. 1983. Age structure, age specific mortality rates and population trend of the freshwater peral mussel (Margaritifera margaritifera) in North Bavaria. - Arch. Hdrobiol. 98:523-532.
Bauer, G. 1987. Reproductive strategy of the freshwater pearl mussel, Margaritifera margaritifera . Journal of Animal Ecology. 56:691-704.
Bauer, G. 1988. Threats to the freshwater pearl mussel Margaritifera margaritifera L. in Central Europe. Biological Conservation. 45:239-253.
Buddensiek, V., G. Ratzbor. 1995. Restoration of sedimetal quality in a small brook of the Lüneburger Heide, Northern Germany by Expertising Bureau for Landscape Engineering, Am Kleinen Felde 17, D-30167 Hannover, Germany Folia Fac. Sci. Nat. Univ. Masarykianae Brunensis, Biologia 91:19-24.
Buddensiek, V. 1991. Untersuchungen zu den Aufwuchsbedingungen der Flußperlmuschel Margaritifera margaritifera Linnaeus (Bivalvia) in ihrer frühen postparasitären Phase. - Thesis, University of Hannover.
Moorkens, E.A. 1999. Conservation management of the freshwater pearl mussel Margaritifera margaritifera. Part 1: Biology of the species and its present situation in Ireland. Irish Wildlife Manuals, No. 8. Dúchas, The Heritage Service, Dubln. 34 pp.
Rensel, J. Analysis of Stream conditions in Upper Bear Creek, Washington. Report prepared for Group Four, Inc., Lynnwood, WA.
Toy, K.A. 1998. Growth, reproduction and habitat preference of the freshwater mussel Margaritifera falcata in Western Washington. Thesis. University of Washington. Seattle, Washington.
Turgeon, D.D., A.E. Bogan, E.V. Coan, W.K. Emerson, W.G. Lyons, W.L. Pratt, C.F.E. Roper, A. Scheltema, F.G. Thompson, and J.D. Williams. 1988. Common and scientific names of aquatic invertebrates from the United States and Canada: Mollusks. American Fisheries Society Special Publ. 16. 277 pp.
Williams, J.D., and Neves, R.J. 1995. Freshwater Mussels: A Neglected and Declining Aquatic Resource. In LaRoe, E.T., Farris, G.S., Puckett, C.E., Doran, P.D., and Mac, M.J. Our Living Resources: a Report to the Nation on the Distribution, Abundance, and Health of US Plants, Animals, and Ecosystems. US Department of the Interior, National Biological Service, Washington DC, United States, p. 362.

 Translate
Translate